Philosophers such as Socrates and philosophical schools such as Stoicism have had a certain influence on psychology and psychotherapy, and thus also on human health. But if philosophy can support human health via psychology, can it not support health more directly, on its own? A growing trend today is to offer philosophical conversations as a […]
Continue readingPage 3 of 50
In a sense, all care strives to be tailored to the individual patient. But the technical possibilities to obtain large amounts of biological data from individuals have increased so significantly that today one is talking about a paradigm shift and a new way of working with disease: precision medicine. Instead of giving all patients with […]
Continue readingWhen ethical dilemmas are discussed using case descriptions or vignettes, we tend to imagine the cases as taken from reality. Of course, the vignettes are usually invented and the descriptions adapted to illustrate ethical principles, but when we discuss the cases, we tend to have the attitude that they are real. Or at least real […]
Continue readingMultiple sclerosis (MS) is a chronic inflammatory disease that affects the nerves of the brain and spinal cord. Symptoms can vary between individuals and the progression of the disease is difficult to predict. Medications can slow the progression of the disease and relieve symptoms, but they do not cure the disease. Symptoms tend to come […]
Continue readingEvery day, researchers receive a motley of offers from dubious journals to publish in them – for a fee. The fact that researchers do not accept these offers does not prevent them from one day discovering that they have become authors of an article that they never wrote. This recently happened to four surprised colleagues […]
Continue readingAuthorities and researchers have a responsibility to the public to disseminate information about risks identified through research. Dissemination of information and education are also an important part of public health efforts to influence norms about lifestyle habits and support individuals in making informed choices about their health. Research has found links between overweight and at […]
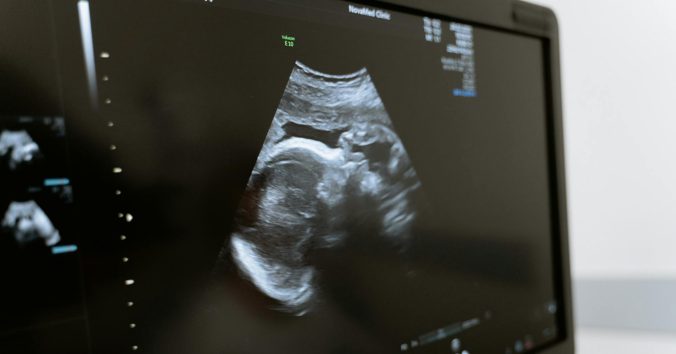
Continue readingA large proportion of pregnant women in Sweden undergo prenatal diagnosis to assess the likelihood of chromosomal anomalies in the fetus. This initially involves a so-called CUB test, which combines ultrasound with a biochemical test. If the CUB test indicates a high probability of a chromosomal anomaly, further tests are offered. This can be a […]
Continue readingHow do you handle a reluctant patient who may not want to take their medication or who protests against measures that are deemed to be beneficial to the patient, such as inserting a urinary catheter? Do you just give up because the patient does not consent? Except in acute emergency situations, coercive somatic treatment is […]
Continue readingMost people are probably aware that antimicrobial resistance is one of the major threats to global health. When microorganisms develop resistance to antibiotics, more people become seriously ill from common infections and more people will die from them. It is like an arms race. By using antibiotics to defend ourselves against infections, we speed up […]
Continue readingWhy is the question of truth so marvelous? A common attitude is that the question can make us check that our opinions really are correct before we express them. By being as well-informed as possible, by examining our opinions so that they form as large and coherent a system as possible of well-considered opinions, we […]
Continue reading